How does pre-implantation genetic screening (PGS) work?
Written by:Having a baby is not always straightforward. Genetics and infertility can conspire to make starting a family seem like an impossibility. However, options such as IVF can help to maximise your chances. One such option is pre-implantation genetic screening (PGS), which can help to streamline the IVF process. Fertility specialist Dr Benjamin Abramov explains.
What is pre-implantation genetic screening (PGS)?
PGS (pre-implantation genetic screening) is a test that can be done during an IVF treatment cycle. It is used to determine which of the embryos are most likely to implant and develop into a baby by examining the chromosome makeup of embryos fertilised via IVF before transferring them into the womb. This knowledge can help to save time and the financial and emotional toll involved with transferring embryos which are destined to fail. However, PGS does not increase the cumulative pregnancy rates.
How does PGS work?
PGS starts off the same as a standard IVF treatment cycle:
- Stimulating egg development
Hormones are used to stimulate a good cohort of eggs to mature – ideally 10-15, although this can vary depending on the individual. Those women with a poorer ovarian reserve will need a higher dose of stimulation drugs than those with a good reserve, and many women will still not have an optimal number of eggs even when high doses of stimulation are used.
- Fertilisation and developing the embryos
The eggs retrieved are then fertilised in the embryology lab. There are two ways of doing this:
- standard IVF procedure – the eggs are simply incubated with sperm overnight
- ICSI (intracytoplasmic sperm injection) – the direct injection of a single sperm cell into an egg. This technique is used when the sperm quality is poor.
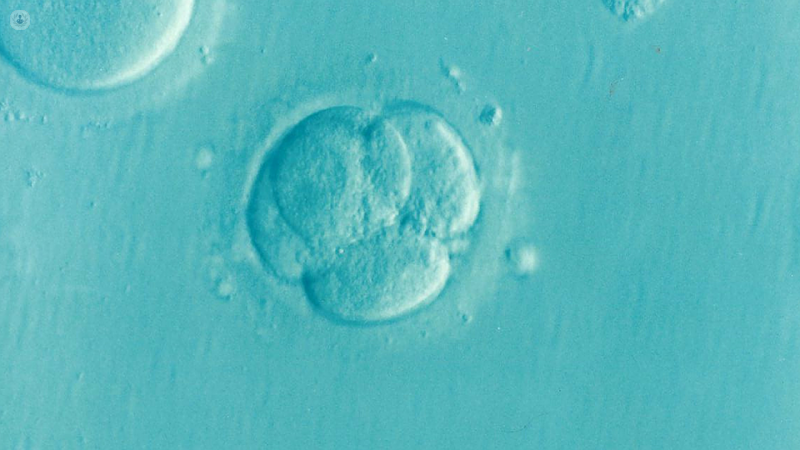
Day 1 (after egg collection): the eggs are checked for signs of normal fertilisation. Normally, fertilised eggs will have 2 small round structures within them called pronuclei. Each represents the male and female genetic material respectively.
Day 2: successfully fertilised eggs will divide into 2-4 cells.
Day 3: eggs divide into 6-8 cells.
Day 4: the embryo consists of 16-32 cells, which are tightly clumped together to form a structure called a morula.
Day 5-6: the embryos develop into a blastocyst form. This is a structured embryo consisting of a ‘shell’ of cells (trophectoderm), which will form the placenta and membranes, a central space filled with fluid (the ‘cyst’) and an inner cell mass. The inner cell mass is destined to form the embryo itself.
- IVF vs. PGS
In standard IVF or ICSI treatment cycles, embryos can be transferred back to the womb at any point after confirming normal fertilisation (after seeing two pronuclei on day 1), the majority occurring between day 2 and day 5 after egg collection.
The idea is to confirm normal development and to ‘race’ the embryos – the embryos that show the best development at certain milestones are more likely to implant and result in a baby. If on day 2 or 3 the ‘winners’ of this ‘race’ are apparent already or the patient has only two good quality embryos, the embryos can be transferred back to the womb.
Yet in many patients, IVF treatment results in more than just two good quality embryos on day 2 or 3, and choosing one or two of them at this stage would be like tossing a coin, so it is worth keeping the embryos in the ‘race’ and incubated in the lab until the blastocyst stage. Blastocysts have a very good chance of implantation and differences in the quality of the embryos at this stage are more telling about their implantation potential.
The process by which embryos are selected on the basis of their development and how they look under the microscope is what we call ‘morphology’ and is the standard tool for embryo selection. Despite this, embryos with a seriously abnormal makeup of chromosomes sometimes appear to be developing well, but will either not implant or may implant and result in a miscarriage.
This is the advantage of using PGS: embryos can be checked for their makeup of chromosomes at any point during the 5-6 days they spend in the lab via a biopsy – a procedure carried out under a microscope to remove some cells from the embryo. The most common current practice is to do the biopsy on day 5 or 6 (blastocyst stage), when it is possible to remove a few cells from the trophectoderm.
These cells are then sent to the genetics lab, where the genetic material is amplified and examined, while the embryos are frozen and stored until we receive the results of the genetic analysis.
When it is confirmed that at least one of the embryos has a normal chromosome makeup, we plan a frozen embryo replacement cycle, during which a chromosomally normal embryo is thawed and transferred into the womb.


