Recent advances in brain tumour surgery
Written by:Professor Keyoumars Ashkan of the London Neurosurgery Partnership is actively involved in research and new developments in neurosurgery, and is the UK Lead for the Genomics England programme for brain tumours. He has published extensively on the subject, and specialises in surgery for the removal of brain tumours. Here, he discusses some of the latest developments, and how they are beneficial for both patient and surgeon.
Removing brain tumours through surgery still remains a challenge, but in recent years a number of technical advances have enabled surgery to become more effective, and safer for the patient.
Intra-operative neuro-navigations
This procedure essentially allows fusion of the patient’s MRI scan obtained before surgery with the anatomy of the patient’s brain on the surgical table. In a way it enables the surgeon to “see” and check their position on the MRI scan as they operate on the brain. The technology increases the precision and accuracy of the surgery, allowing for smaller incisions, increasing surgical safety as well as facilitating quicker recovery.
Fluorescence guided resection
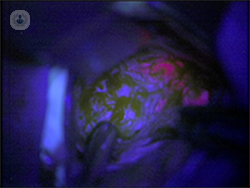
Certain tumours can ‘take in’ specific drugs which are administered to the patient before the operation, converting them into a fluorescent compound. The tumour is then viewed through a special type of microscope in theatre, where it can be seen fluorescing thanks to the administered drug. The tumour shows up as red, while the rest of the brain looks blue, allowing the surgeon to distinguish the tumour from the normal brain, and meaning a more complete resection is easier to achieve.
Intra-operative chemotherapy
Many patients with aggressive tumours require systemic chemotherapy (given orally or by injection) as part of their treatment. This is often commenced a few weeks after the surgery to allow for the wounds to heal. During this interval, any residual tumour is likely to continue to grow. Intra-operative chemotherapy acts to bridge this gap. It is now possible to line the cavity left behind after the surgery with intra-operative chemotherapy compounds, which can help damage the residual tumour cells whilst the patient recovers enough to start systemic chemotherapy.
Brain mapping
Certain tumours may be located in close proximity to the critical areas of the brain such as those responsible for movement or speech. It is therefore critical to know the exact anatomical relationships between the tumour and these eloquent brain areas. This is called brain mapping. It is now possible to do this with a high degree of accuracy pre-operatively. Certain advanced MRI techniques such as functional MRI and tractography can be very helpful here. Trans-cranial magnetic stimulation (TMS), however, is now probably leading the way.
The TMS technique uses a special magnet to allow non-invasive mapping of the brain before surgery in the comfort of an examination room over a short time. Thus the surgeon can have invaluable knowledge beforehand on what to expect when they perform the operation, both aiding the informed consent process when counselling the patient, and of course planning of the surgery. With patients in whom pre-operative mapping has identified potential risks due to the very close proximity of the tumour to the critical brain areas, it is now also possible to map the brain during the surgery to millimetre accuracy. For patients with tumours near the motor parts of the brain, intra-operative mapping can be done under general anaesthetic, whilst in patients with tumours near the speech parts of the brain, mapping is done with the patient awake during the surgery under local anaesthetic (awake craniotomy). Brain mapping is now allowing us to safely tackle many tumours previously deemed inoperable or too dangerous to operate on.
Robotic surgery
Robotic surgery is being used across many specialties nowadays, but it is now beginning to find its place in neurosurgery and brain tumour surgery. Robotic surgery allows the surgeon to operate with an incredible level of accuracy, which is especially useful in approaching deep seated brain tumours. Accuracy is critical for these types of tumours, which can now be reached all the way from the surface to the depth, allowing a diagnostic biopsy to be performed.
Visualsae
This technology combines robotics with laser surgery to allow surgeons to treat deep seated tumours where previously only a biopsy would have been possible, given the risk surgery carries to reach these areas. The robot allows accurate placement of the fine laser delivery tube into the tumour. The laser is then used to heat up and damage the tumour under real time MRI guidance which also allows for the measurement of the temperature to avoid any damage to the surrounding brain. The technology provides a novel therapy approach for many previously-thought inoperable deep primary or secondary (metastatic) brain tumours.
Radiosurgery
This is the use of focused radiation to treat tumours. Although strictly speaking not surgery, the treatment is often planned by surgeons, as a critical knowledge of the anatomy is needed. Gamma-knife radiosurgery is an accurate, specialised treatment, which uses intersecting beams of gamma radiation to treat the tumour. Gamma-knife radiosurgery requires the patient to have their head fixed into a frame during the procedure. The more recent technology of Cyberknife radiotherapy does not require this by virtue of using an intelligent head tracking system, thus increasing the comfort of the therapy for the patients. The most common use of radiosurgery is for patients with multiple brain metastasis. Some benign brain tumours such as meningiomas or acoustic neuromas are also particularly suited to radiosurgery.
To find out more about Professor Ashkan and the London Neurosurgery Partnership click here